
This shrinks the size of the atom then the atomic radius is reduced. Therefore, the attractive force between electrons and the nucleus is high. But moving along a period increases the number of protons in the nucleus along with the number of electrons present in the atom. Therefore, the atom is large and the atomic radius is also large. At the beginning of the period there are fewer electrons in the outermost orbital. 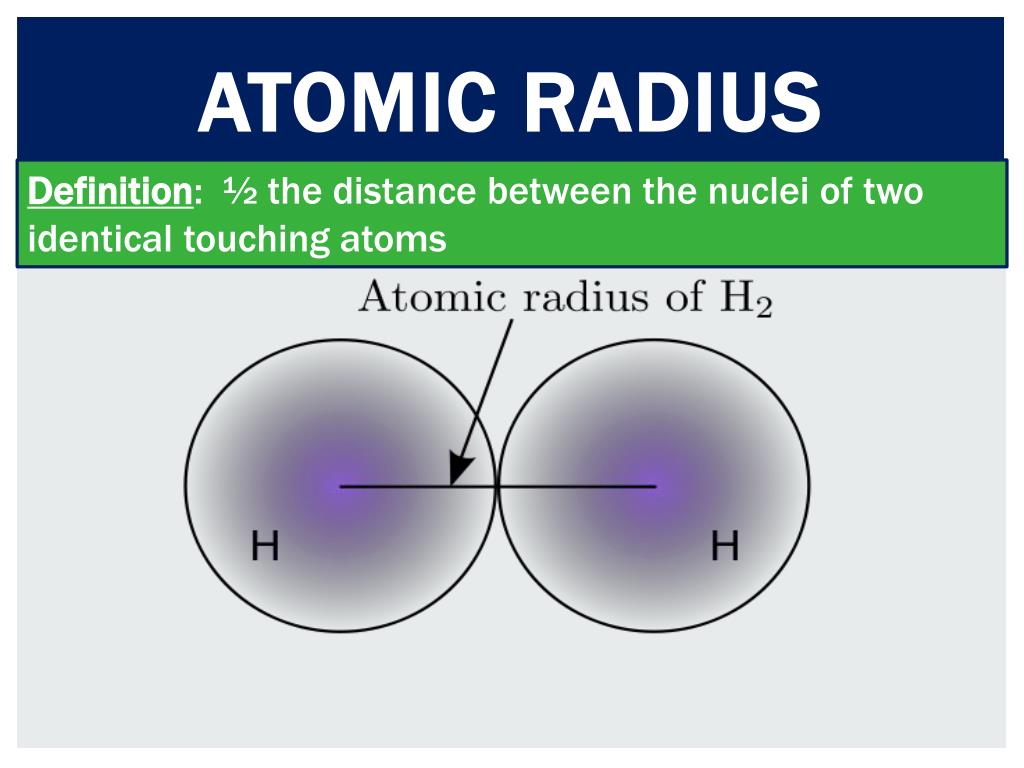
When the number of electrons present is higher, the attraction between the electrons and the nucleus is also high. The elements in the same period have the same number of electron shells. The atomic number gradually decreases along a period of the periodic table. When looking at the Periodic Table of the Elements there is a pattern of the atomic radius of the elements. Atomic radius can only be defined for isolated and neutral atoms. 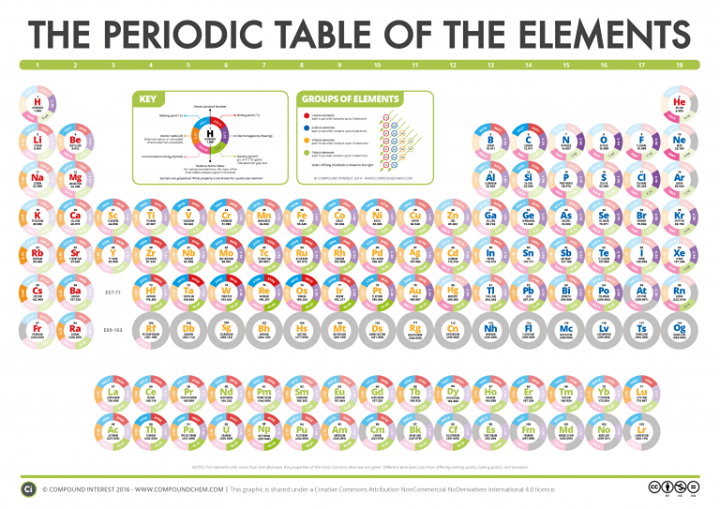
In other words, it is the distance from the nucleus to the most distant electron that belongs to that atom. The atomic radius is the distance from the atomic nucleus to its limit of the electron cloud. Key terms: atomic radius, atoms, electron shell, ionic radius, ions What is atomic radius? What is the difference between atomic radius and ionic radius - comparison of the main differences What is ionic radius - definition, trends in the periodic table 3. What is atomic radius - definition, trends in the periodic table 2. The main difference between atomic radius and ionic radius is that the atomic radius is the radius of a neutral atom while the ionic radius is the radius of an electrically charged atom. The radius of an ion can be either larger or smaller than the radius of an atom, depending on the electrical charge of the ion. The ionic radius is the radius of the ion of an atom. The atomic radius is the distance between the nucleus of an atom and the boundary of its electron cloud. Because an atom or ion consists of electrons that are in motion. Since atoms and ions are circular 3D structures, we can measure the radius of an atom or an ion. These atoms can be converted into ions by adding one or more electrons from outside. Key difference - atomic radius vs ionic radiusĪtoms are the building blocks of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
